This post is part of our 2017 Summer of Science series! Here you’ll find quick and easy projects and experiments you can do this summer to keep your kids’ brains active and curious! As always, you can go as much or as little into the science explained below based on age and interest. Enjoy!

This project actually came about kind of by accident, but honestly that’s how lots of science discovery happens! I was washing the extract of turmeric from a bowl in my sink (we were making dye for tie-dye!) and realized the color changed from bright yellow to brownish-red when I added water. I googled the color change and found that curcumin, the main compound responsible for turmeric’s color, will turn red in solutions with pH greater than 7.5. I took out my (real) pH strips and clocked the pH of my tap water at 9. After making sure that it’s not big deal to drink pH 9 tap water, I decided to see if we could make some pH strips with the turmeric extract, and it worked splendidly!
You will use isopropanol (rubbing alcohol) to make a crude turmeric extract that contains a lot of the compound curcumin. This molecule is responsible for the color change in the pH strips. Ethanol will also work, but water will not, as curcumin is not very soluble in water.
This is a fun project for kids, because (besides the beautiful colors) you actually get to make an item that can be used for practical purposes. After you make the strips, try them out on various household solutions in your fridge or pantry and see if they are acidic or basic!
Below you’ll find the easy procedure to make your own pH strips and also a quick lesson about pH and how curcumin’s color changes!
Acids and Bases
Hydrogen is the smallest atom. It has one proton and one electron. When it is part of a molecule, it shares that electron with the atom it is attached to. In some molecules, when they are dissolved in water, the hydrogen will leave the molecule, also leaving behind it’s electron. This makes the hydrogen just a proton now! A molecule that does this is called an acid. If a another molecule can take up that proton floating around, it is called a base.
Put another way, acids are compounds that are likely to donate a proton and bases are compounds likely to accept a proton. This happens because certain electrons on the base (called “lone pair electrons”) are more attracted to the proton than the lone pair electrons that were interacting with the proton on the acid. In other words, an acid/base reaction happens when the base “wants” the acid’s proton more than the acid does, so it basically (haaa!) gets ripped away. When you get a strong acid or base on your skin, it’s this forceful donation or ripping of protons from the proteins and lipids in your skin that causes a burn. If this happens too much, the proteins can begin to lose their shape and function. Most proteins cannot get their shape or function back if disturbed, so this basically is what causes the damage to your skin.
But all acid/base reactions aren’t so scary. There are some very peaceful ones where the molecules playfully pass protons, like in the case of curcumin (the acid in this case) and a base.
When a base touches your pH strips, the base takes a proton from curcumin, the main source of the yellow-orange color in turmeric. The deprotonated curcumin is red because the missing proton changes how electrons are distributed throughout the molecule, which changes the wavelengths of light it absorbs. Different wavelengths of light, in this case, translate to different colors. It’s amazing the disturbance one little proton can cause!
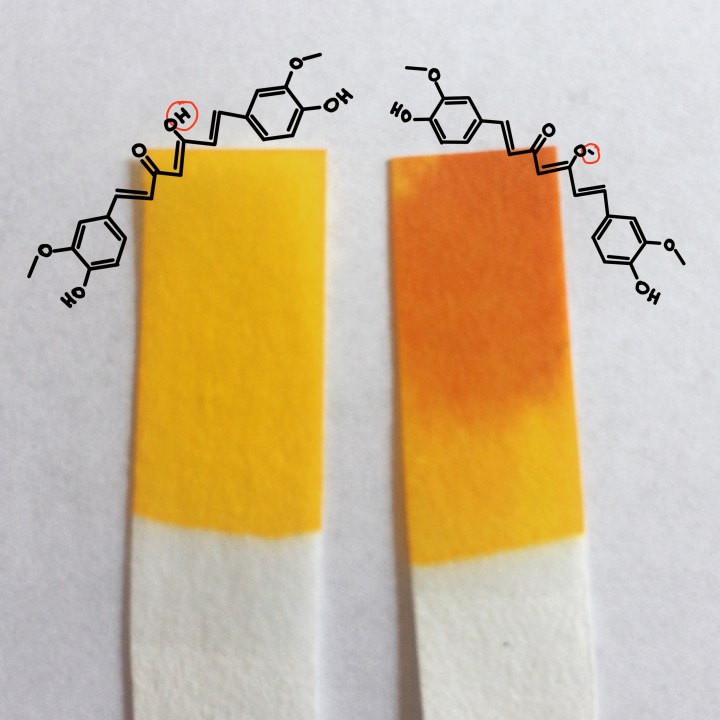
pH tells us how acidic a solution is. A low pH means a solution is more acidic (higher concentration of protons) and a high pH means a solution is more basic (higher concentrations of molecules willing to take a proton). A neutral solution has a pH of 7 and this means the amount of acids equal the amount of bases. Curcumin changes color (or loses that proton) around pH 7.5-8, so at a slightly basic pH.
Making the Curcumin pH Strips
Warnings: Even though food products are used, you should not eat anything from this project. Isopropanol is poisonous if ingested, so do not drink the turmeric extract, and isopropanol is flammable, so do not work near an open flame. Work in a well-ventilated area. You should also wear goggles to protect your eyes. The turmeric extract will stain fingers and clothes, so wear gloves, unimportant clothes, and work on a surface you wouldn’t mind if it turned yellow forever. When using your pH strips with kids, don’t let them test them on strong or volatile acids or bases (lye, Draino, bleach, ammonia, etc.), it’s just not worth the risk. Safer examples are listed below.
Materials
(This section uses affiliate links. If you click on the link and make a purchase, we get a share of the price to help keep this blog running! Thank you!)
-
- Turmeric
- Isopropyl Alcohol/Isopropanol
(91% or 99%) (aka Rubbing Alcohol)
- glass jars and something to cover it (lid, coaster)
- measuring spoons
- Filter Paper
/ Coffee filters
- Turmeric
- household products to use your strips (examples listed below)
Method
 Add 2 TBS of isopropanol to a glass jar
Add 2 TBS of isopropanol to a glass jar- Mix in 1 tsp of turmeric
- Let sit, mixing every now and then, for 10 min. (Or you can leave it for a few hours). Make sure to cover it, isopropanol evaporates quickly.
- Meanwhile, cut your filter paper/ coffee filters into strips (~1/4-1/2 inch x 2 inches)
- Filter the liquid from the leftover turmeric by letting
it settle, then carefully pouring the top liquid layer through a new piece of filter paper or coffee filter, then collecting the clear yellow-orange liquid that flows through.
then collecting the clear yellow-orange liquid that flows through. - Quickly dip your cut paper strips into the clear yellow orange liquid to about half way up the paper (~1 inch). Blot off excess. Alternately, you can lightly paint it on the strips with a paintbrush.
- Let dry on a non-porous surface.
- To use your pH strips, pour a small sample of your liquid to test into a cup or jar (never dip strips into the main vessel, this will contaminate the stock). Dip the
 strip in and swirl around for about 10 seconds. Take out the strip and observe the color!
strip in and swirl around for about 10 seconds. Take out the strip and observe the color!
Example of liquids to test:
- Vinegar
- 1/4 tsp baking soda in 1/4 cup water (you can eyeball the amounts)
- lemonade/ lemon or lime juice
- milk
- soapy water
- tap water
- milk of magnesia
- ocean or lake water
*Note: if you don’t have filter paper or coffee filters for the strips, you can also use pieces of a white natural fabric, like cotton.
Example Activity
 With my 2.5 year old daughter, we tested vinegar and the baking soda solution with out strips. Afterwards, I gave her an eyedropper and let her drop vinegar into baking soda (powered form) for fun. But then when she finished, she asked if she could use one of the strips to test the “goopy” (the slurry left over from mixing the vinegar and baking soda)! I was so proud! So we did, and luckily (I didn’t measure out the vinegar and baking soda, so I wasn’t sure how much excess there would be of one of them) the strip ended up being an orange color in between the vinegar’s yellow strip and baking soda’s red strip. Not a perfect quantitative experiment (if there were equal amounts acid and base reacting, the solution would’ve been neutral, resulting in a yellow test strip in this case), but very illustrative for young ones, and I was so happy to see her curiosity and understanding that what she made was a new “goopy” now and needed to be tested!
With my 2.5 year old daughter, we tested vinegar and the baking soda solution with out strips. Afterwards, I gave her an eyedropper and let her drop vinegar into baking soda (powered form) for fun. But then when she finished, she asked if she could use one of the strips to test the “goopy” (the slurry left over from mixing the vinegar and baking soda)! I was so proud! So we did, and luckily (I didn’t measure out the vinegar and baking soda, so I wasn’t sure how much excess there would be of one of them) the strip ended up being an orange color in between the vinegar’s yellow strip and baking soda’s red strip. Not a perfect quantitative experiment (if there were equal amounts acid and base reacting, the solution would’ve been neutral, resulting in a yellow test strip in this case), but very illustrative for young ones, and I was so happy to see her curiosity and understanding that what she made was a new “goopy” now and needed to be tested!
Questions to Ponder
- Can these strips tell you if something is definitely an acid or a base? (Hint: What pH is neutral and at what pH do these strips change color?)
- Did some of your test strips turn back to yellow after turning red or orange? Why do you think this happens?
- Look at the three strips below. What are some possible reasons why we can see three colors, when I only talk about one deprotonation event above?

I hope you have fun doing this project! Let me know how it goes! And keep an eye out for another post on how to use your leftover turmeric extract!

