
This post is part of our Summer of Science series! Check back periodically to find more fun, easy, and affordable experiments and projects you can do at home with your kids this summer. This project is about the enzyme, bromelain, which degrades other proteins. You will use it to turn chicken to mush while learning about experimental set up, controls, and the wonderful world of enzymes.
Oh, the first time I learned about bromelain was in college on a trip to Hawaii (I got a chance to observe Mars on the NASA IRTF on Mauna Kea). My friends and I flew there a week earlier than our telescope time to explore the islands a bit. One day, we each bought a fresh pineapple and each ate. the. whole. thing. Never do this, it was awful. I mean it tasted absolutely delicious, but our tongues and mouths were sloughing skin off for a few days as they recovered from being eaten themselves by the enzyme, bromelain.
Pineapple fruit has a very high concentration of fruit bromelain, a protein that can break down other proteins, aka a “proteolytic enzyme”.
Pineapple stem also has a high concentration of stem bromelain. People have long used these enzymes for health applications like wound debridement, and there are currently many (…dubious) claims of miracle cure status. Another use lies in the culinary realm, which became quite obvious after the state of my tongue that day: a meat tenderizer. You can pour some pineapple juice on chicken or steak, or include it in a marinade to make tough meats softer before cooking. But don’t store pineapple with other meats! I also know from experience that the meat turns to mush as the enzymes work their magic overnight in your fridge. This is similar to what we did in the experiment: watching and feeling bromelain break down meat, and experimenting with different conditions to see how it affects the activity of the enzyme.
You can also buy a purified form of bromelain (most likely stem bromelain, but possibly a mix of the two) aptly called “Meat Tenderizer” at almost any grocery store. That’s what we used in the experiments. Feel free to use fresh pineapple juice, but using the free enzyme in Meat Tenderizer allows you the flexibility to teach about the importance of different buffers and salt concentrations on enzyme activity. If you use pineapple juice, be sure to use it fresh from the fruit. The enzyme will be inactive in pasteurized bottles and canned juice due to the heat involved in the preparative processes. In fresh juices bought at the store, the enzyme may not be as active as if you fresh squeezed it yourself because enzymes lose activity over time.
A Little Information About Enzymes
Enzymes are large molecules, a special class of proteins that are in all living things. They can speed up or “catalyze” reactions between other molecules. Many important reactions in our bodies would take way too long to occur if nothing was helping them along. We have enzymes to not only speed up these reactions, but also to make sure the correct molecules are reacting. Every reaction in our body, except a small handful, is controlled by an enzyme.
The reaction being catalyzed here is called “amide hydrolysis.” An amide is a type of chemical bond. It used to hold the building blocks of proteins (amino acids) together. It is a very stable bond, which is why living things need an enzyme to break it. Without an enzyme, it takes hundreds of years for half the amide bonds in solution to split. With an enzyme, it takes less than a second. The term “hydrolysis” in amide hydrolysis refers to water being used to break the bond. This term is a little misleading here if you’re not familiar with organic chemistry, but just know every hydrolysis reaction adds the atoms of a water molecule to the molecules being split. Water, however, in this case does not break the amide bond, it is the interplay of molecules on the enzyme (sometimes with water) that break it.
All enzymes need to be in certain conditions to work. Enzymes in your stomach acid work their best at a low pH, because your stomach acid has a low pH. Enzymes in your saliva work best around a neutral pH, because your saliva has around a neutral pH. When you swallow your saliva, the saliva enzymes become inactive in your stomach acid because the pH is too low. Likewise if you tried to make enzymes from your stomach acid work at a neutral pH, they wouldn’t be able to function well. These concepts also apply when you’re working with enzymes in the lab (or in your kitchen). You need the correct conditions to allow the enzyme to work to the best of its abilities. Bromelain works the best at acidic pH, around pH 3 (scientists have found this out by seeing how fast it works at a range of pH values).
In this project, we used lemon-lime Gatorade to bring the pH of our water down to 3 (we have pH strips to test this) and then dissolved the Meat Tenderizer Bromelain into the Gatorade solution. You can also use pasteurized pineapple juice and Meat Tenderizer (because pineapple juice is the solution it evolved to work in, after all!). Alternately, you can use freshly squeezed pineapple juice which will have the correct conditions and enzymes all in one. I recommend using Meat Tenderizer and Gatorade/juice because having kids add multiple things together stresses the importance of each component, and allows you to play around with other variables.
Materials
(Affiliate links)
- Non Seasoned Meat Tenderizer
- Lemon-lime Gatorade or pasteurized pineapple juice (you can substitute this and the meat tenderizer for freshly squeezed pineapple juice)
- Cooked pieces of meat or other protein, we used chicken
- Bowls or plates
- Optional: pH strips
with 1 pH unit resolution
- Optional: heating pad
- Optional: refrigerator
Procedure
You can be as exact or ball park as you want in this project, depending on what you want to get out of it. If you want to treat it as an experiment, measure everything! Mass of meat tenderizer, volume of liquids, temperature. But if you just want to see enzymes at work, sprinkle on some meat tenderizer and squish away. The procedure below is a happy medium of those options.
- Dilute the Gatorade by half (i.e. 1/2 cup water + 1/2 cup Gatorade)
- Cut pieces of chicken to about 1/4 inch x 1 inch x 1 inch, give or take. Just try to make sure the chicken is thin, and that the pieces are the same size and thickness.
- Take 1/4 cup of Gatorade and add 1/4 tsp of Meat Tenderizer, and swirl until it dissolves.
- Find two bowls, cups, watch glasses, saucers, curved plates…anything that will hold the Gatorade solution close to the chicken.
- Pour the Meat Tenderizer/ dilute Gatorade solution into one container, and only dilute Gatorade in the other. Mark which one is which! 🙂
- Gently place a piece or two of chicken into each solution.
- Wait a few hours (or you can vary the time), then start gently squishing your meat. Do they feel different? Do they look different before or after you squish it?
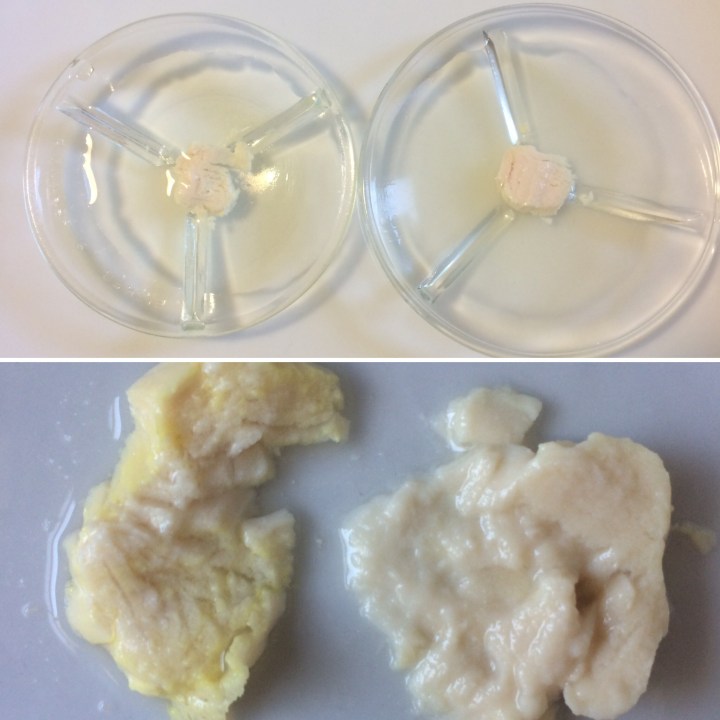
Our chicken sample during (top) and after (bottom) partial hydrolysis by bromelain! On the left, the sample had no enzyme in solution, and on the right, bromelain was included. Can you see the difference between the samples in the bottom? This was after pressing each with my finger.
Alternate Procedures
- Try changing the temperature of the samples above, by putting two (with and without enzyme) into the refrigerator, two at room temperature, and two on a heating pad or in a warm place. Compare all six!
- Try putting in more or less Meat Tenderizer and compare the effect of different concentrations of enzymes.
- Try using doing it in just water. Does it still work?
- Trying adding a lot of salt to the solutions. What happens?
- Try degrading different types of meat of different types of protein and compare the results. Here, we used gelatin! It worked great, and there was a huge difference between the sample that had enzyme (right) and did not (left).

- This project is great to show the importance of controls. You might even try one, intentionally, without a control and use the results to discuss how you can’t make any assumptions without appropriate controls. For example, if you left out the control in the main project above, could you conclude that the enzyme made it mushy, or was it just from sitting in the gatorade for a few hours? You wouldn’t know unless you did a “no enzyme” control. Making “mistakes” is often one of the best ways to learn.
Let me know how this project goes! Hope you all have fun!
Sources:
Bromelain enzymes. Takashi Murachi. Methods in Enzymology, Volume 45, 1976, pp 475-485


Lovely postt
LikeLike
Is there a way to quantitatively measure the effect of bromelein on the gelatin?
LikeLike
There would be several possible ways in a laboratory setting, but none at home (that I’m aware of!). In what context were you curious about?
LikeLike